Prakash Lab

Welcome to the Prakash Lab at UCSF.
We are an immunology lab focused on innate immune mechanisms and inflammation seen in critical illness.
We work closely with the Hellman Laboratory also in the Department of Anesthesia and Perioperative Care at UCSF.
We are interested in the interface between sterile and infectious inflammation as it pertains to patients with severe trauma, hemorrhage, and complicated hospital courses.
We are a young lab that is growing and expanding.
San Francisco and the Bay Area in general is a wonderful place to live and work. The scientific community at UCSF is unparalleled.


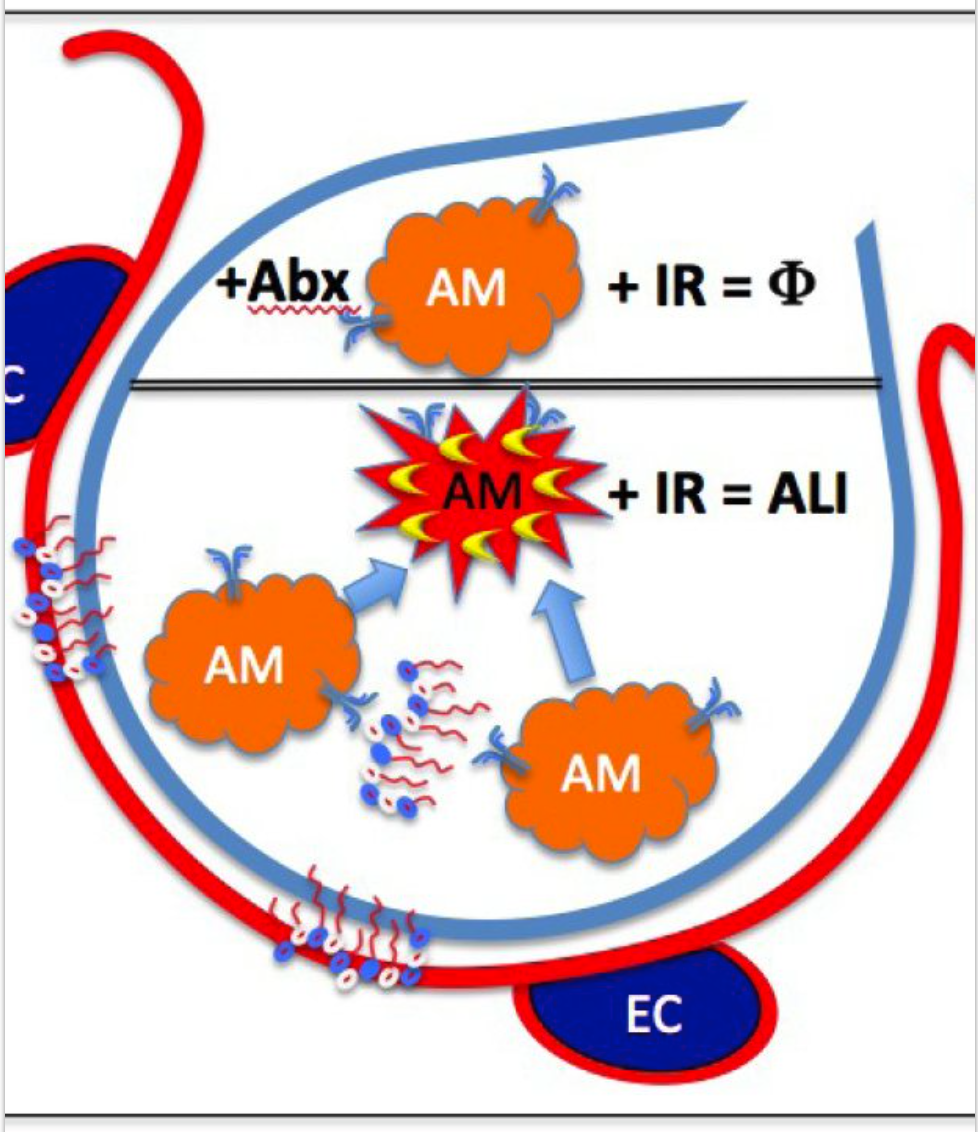
Our primary research focus is broadly on the interface between sterile and infectious inflammation and how the innate immune system is able to distinguish between the two and respond appropriately. Given my clinical interest in trauma anesthesia, and specifically the airway, respiratory and hemodynamic management of critically-ill trauma patients, the lab's research focuses on lung injury in trauma and the pathophysiology of lung ischemia reperfusion (IR) injury. We are interested in how damage markers are generated in IR injury, how these are sensed and how infiltrating immune inflammatory cells are targeted to the lung. Dissecting the individual aspects of these processes will provide important insights towards improving the way trauma and other patients with IR injury are clinically cared for.
Furthermore, by understanding the molecular and cellular basis of lung IR injury, we hope to gain insights into how this process interfaces with coexisting or superimposed infections that are encountered by sick trauma patients in the ICU. These pathogenic, commonly bacterial, nosocomial infections often target the lung with devastating consequences given the relative immune paralysis that often accompanies severe trauma.
We utilize a mouse model of lung ischemia reperfusion injury to investigate the details of this maladaptive process. By using fluorescent and bioluminescent markers to track the presence and activation status of inflammatory cells in live animals, we can determine the kinetics of the initiation, progression, and resolution phases of the lung IR-generated inflammation, as well as the effects of therapeutic interventions. We also aim to use fluorescently tagged bacteria as well to track active infections and identify the primary infected/sensor cell type(s). Understanding the interactions between lung IR injury and concurrent lung infection would allow us to design treatment and prevention strategies for severely ill hospitalized trauma patients.
Mouse Model of Lung Ischemia Reperfusion (IR)
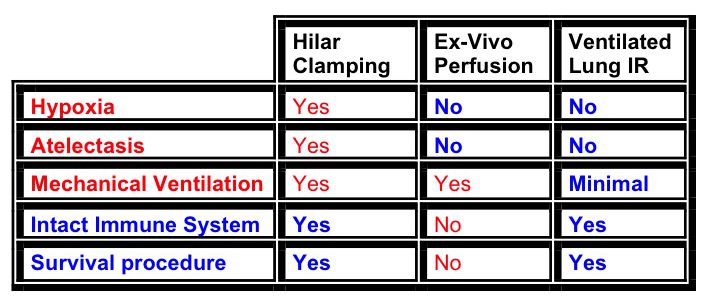
What is key about our model of lung ischemia reperfusion (ventilated lung IR) is that oxygenation and gas exchange continues throughout the process with minimal periods of mechanical ventilation. Other factors that make this model superior to other existing models of lung IR are shown to the right.
This unilateral ventilated lung ischemia reperfusion mouse model was developed and refined by Jeffrey Dodd-o from Johns Hopkins Medicine: Department of Anesthesia and Critical Care Medicine.
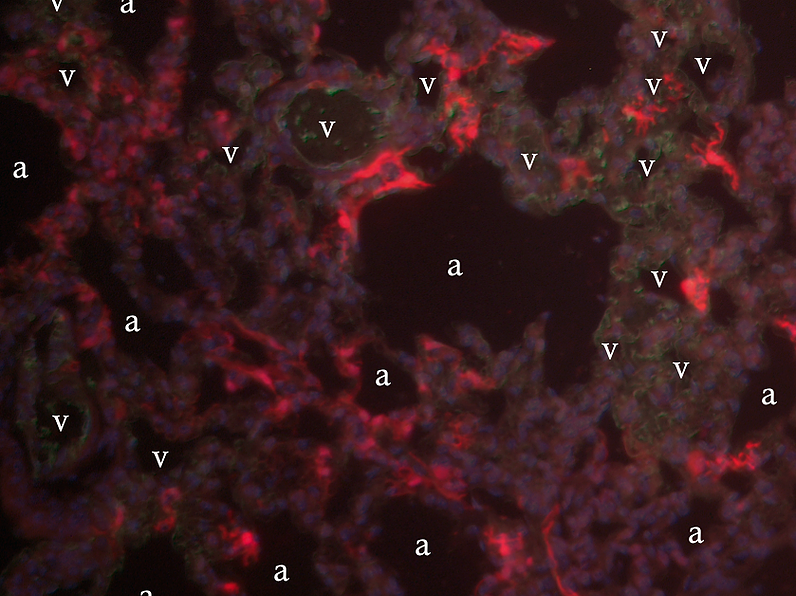
Neutrophils trafficking to the lung after ischemia reperfusion injury are not activated and as shown above are localized to the lung interstitium (red fluorescent signal = RFP+ neutrophils) and not in the alveoli (a) or vasculature (v)

Marié I, Smith EJ, Prakash A, Levy DE.(2000). Phosphorylation-induced dimerization of interferon regulatory factor 7 unmasks DNA binding and a bipartite transactivation domain. Molecular and Cellular Biology, 20(23):8803-14.
PubMed abstract
PDF Version
Smith EJ, Marié I, Prakash A, Garcia-Sastre A, Levy DE. (2001). IRF3 and IRF7 phosphorylation in virus-infected cells does not require double-stranded RNA-dependent protein kinase R or Ikappa B kinase but is blocked by Vaccinia virus E3L protein. Journal of Biological Chemistry, 276(12):8951-7.
Journal Article
PDF Version
Levy DE, Marié I, Smith EJ, Prakash A. (2002). Enhancement and diversification of IFN induction by IRF-7-mediated positive feedback. Journal of Interferon and Cytokine Research, 22(1):87-93.
Journal Article
PDF Version
Caillaud A, Prakash A, Smith EJ, Masumi A, Hovanessian AG, Levy DE, Marié I. (2002). Acetylation of interferon regulatory factor-7 by p300/CREB-binding protein (CBP)-associated factor (PCAF) impairs its DNA binding. Journal of Biological Chemistry, 277(51):49417-21
Levy DE, Marié I, Prakash A. (2003). Ringing the interferon alarm: differential regulation of gene expression at the interface between innate and adaptive immunity. Current Opinions in Immunology, 15(1): 52-8
Abstract
PDF Version
Prakash A, Smith EJ, Lee CK, Levy DE. (2005). Tissue-specific positive feedback requirements for production of type I interferon following virus infection. Journal of Biological Chemistry, 280(19):18651-7
PubMed Abstract
PDF Version
Prakash A, Levy DE. (2006). Regulation of IRF7 through cell type-specific protein stability. Biochemical and Biophysical Research Communications, 342(1):50-6.
PubMed Abstract
PDF Version
Prakash A and Hellman J. (2010). Pattern Recognition Receptors and Factor B: “Complement”ary Pathways Converge. Journal of Leukocyte Biology, 88(4):605-7.
Shin H-S, Xu F, Bagchi A, Herrup E, Prakash A, Valentine C, Kulkarni H, Wilhelmsen K, Warren S, Hellman J. (2011). Bacterial Lipoprotein TLR2 Agonists Broadly Modulate Endothelial function and Coagulation Pathways In Vitro and In Vivo. Journal of Immunology, 186(2):1119-30.
Wilhelmsen K, Mesa KR, Prakash A, Xu F, Hellman J. (2011). Activation of endothelial TLR2 by bacterial lipoprotein upregulates proteins specific for the neutrophil response. Innate Immunity, Dec 20 [Epub ahead of print].
Journal Article
PDF Version
Prakash A, Mesa KR, Wilhelmsen K, Xu F, Dodd-o JM, Hellman J. (2012). Alveolar macrophages and TLR4 mediate lung ischemia reperfusion injury. Anesthesiology, 117(4):822-835.
Abstract
PDF Version
Breyer KEW, Ou J, Durack JC, Prakash A. (2012). Prolonged profound hypoxia and cardiac failure in a young female presenting to the emergency department: unexpected pulmonary embolus, catheter-directed thrombolysis, and subsequent multi-organ failure. ICU Director, 3(5):215-219.
Journal Article
PDF Version
Zhang N, Francis KP, Prakash A, Ansaldi D. (2013). Enhanced detection of myeloperoxidase activity in deep tissues through luminescent excitation of near-infrared nanoparticles. Nature Medicine, 19(4):500-5.
Abstract
PDF Version
Wilhelmsen K, Xu F, Farrar K, Tran A, Khakpour S, Sundar S, Prakash A, Wang J, Gray NS and Hellman J. Extracellular Signal-Regulated Kinase 5 Promotes Acute Cellular and Systemic Inflammation. Science Signaling 2015, Aug 25; vol.8 (391): pp. ra86.
PDF Version
Kangelaris KN, Prakash A, Liu KD, Aouizerat B, Woodruff PG, Erle DJ,Rogers A, Seeley EJ, Chu J, Liu T, Osterberg-Deiss T, Zhuo H, Matthay MA, Calfee CS. Increased expression of neutrophil-related genes in patients with early sepsis-induced ARDS. Am J Physiol Lung Cell Mol Physiol. 2015 Jun 1; 308(11):L1102-13. PMID: 25795726. PMCID: PMC4451399
Prakash A, Sundar SV, Zhu YG, Tran A, Lee JW, Lowell C, Hellman J. Lung Ischemia-Reperfusion is a Sterile Inflammatory Process Influenced by Commensal Microbiota in Mice. Shock. 2015 Sep; 44(3):272-9. PMID: 26196836. PMCID: PMC4537678
PDF Version
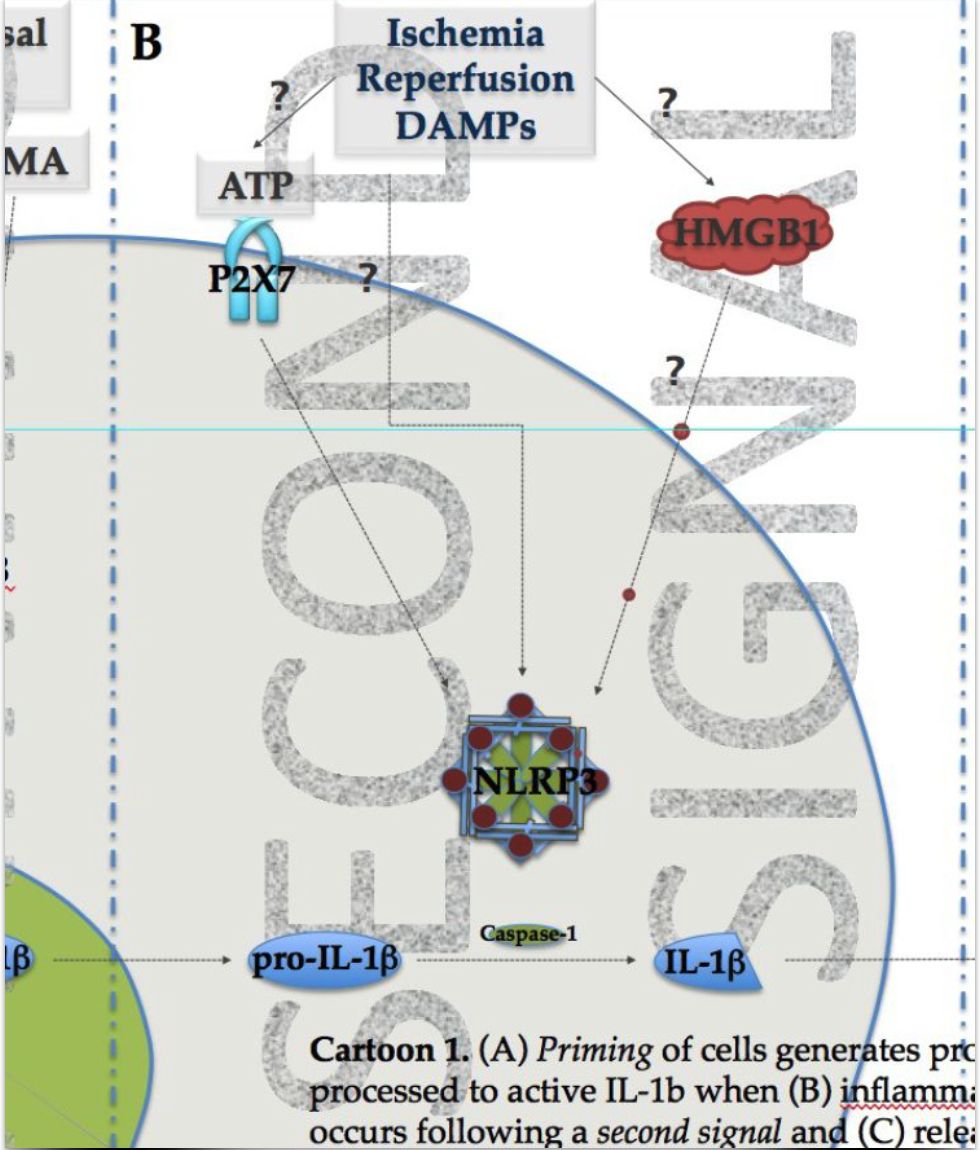
Tian X, Sun H, Casbon AJ, Lim E, Francis KP, Hellman J, Prakash A. NLRP3 Inflammasome Mediates Dormant Neutrophil Recruitment following Sterile Lung Injury and Protects against Subsequent Bacterial Pneumonia in Mice. Front Immunol. 2017; 8:1337. PMID: 29163464.
PDF Version
Lawton SK, Xu F, Tran A, Wong E, Prakash A, Schumacher M, Hellman J, Wilhelmsen K. N-Arachidonoyl Dopamine Modulates Acute Systemic Inflammation via Nonhematopoietic TRPV1. J Immunol. 2017 Aug 15; 199(4):1465-1475. PMID: 28701511. PMCID: PMC5544930
Tian X, Sun H, Casbon AJ, Lim E, Francis KP, Hellman J, Prakash A. NLRP3 Inflammasome Mediates Dormant Neutrophil Recruitment following Sterile Lung Injury and Protects against Subsequent Bacterial Pneumonia in Mice. Front Immunol. 2017; 8:1337.
PubMed
Tian X, Hellman J, Horswill AR, Crosby HA, Francis KP, Prakash A. Elevated Gut Microbiome-Derived Propionate Levels Are Associated With Reduced Sterile Lung Inflammation and Bacterial Immunity in Mice. Front. Microbiol. 10:159. doi: 10.3389/fmicb.2019.00159
PDF Version
Xiaoli Tian, He Sun, Amy-Jo Casbon, Edward Lim, Kevin P. Francis, Judith Hellman, and Arun Prakash. NLRP3 Inflammasome Mediates Dormant Neutrophil Recruitment following Sterile Lung Injury and Protects against Subsequent Bacterial Pneumonia in Mice. published: 31 October 2017 doi: 10.3389/fimmu.2017.01337
PDF Version
Kirsten Neudoerffer Kangelaris, Arun Prakash, Kathleen D. Liu, Bradley Aouizerat, Prescott G. Woodruff, David J. Erle, Angela Rogers, Eric J. Seeley, Jeffrey Chu, Tom Liu, Thomas Osterberg-Deiss, Hanjing Zhuo, Michael A. Matthay, and Carolyn S. Calfee. Increased expression of neutrophil-related genes in patients with early sepsis-induced ARDS. First published March 20, 2015; doi:10.1152/ajplung.00380.2014
PDF Version
Samira K. Lawton, Fengyun Xu, Alphonso Tran, Erika Wong, Arun Prakash, Mark Schumacher, Judith Hellman, and Kevin Wilhelmsen. N-Arachidonoyl Dopamine Modulates Acute Systemic Inflammation via Nonhematopoietic TRPV1. J Immunol 2017; 199:1465-1475; Prepublished online 12 July 2017; doi: 10.4049/jimmunol.1602151
PDF Version
Tian X, Hellman J, Horswill AR, Crosby HA, Francis KP, Prakash A. Corrigendum: Elevated Gut Microbiome-Derived Propionate Levels Are Associated With Reduced Sterile Lung Inflammation and Bacterial Immunity in Mice. Front Microbiol. 2019; 10:518.
PubMed
Kornblith LZ, Bainton CMV, Fields AT, Matthay ZA, Magid NT, Nunez-Garcia B, Prakash A, Kurien PA, Callcut RA, Cohen MJ, Bainton RJ. A journey upstream: Fluctuating platelet-specific genes in cell-free plasma as proof-of-concept for using ribonucleic acid sequencing to improve understanding of postinjury platelet biology. J Trauma Acute Care Surg. 2020 06; 88(6):742-751.
PubMed
Won Lee, Steven Straube, Ryan Sincic, Jeanne A Noble, Juan Carlos Montoy, Aaron E Kornblith, Arun Prakash, Ralph Wang, Roland Bainton, Philip Kurien. Clinical Evaluation of a COVID-19 Antibody Lateral Flow Assay using Point of Care Samples. J Trauma Acute Care Surg. 2020 06; 88(6):742-751.
PubMed
Liu Q, Tian X, Maruyama D, Arjomandi M, Prakash A. Lung immune tone via gut-lung axis: gut-derived LPS and short-chain fatty acids' immunometabolic regulation of lung IL-1β, FFAR2, and FFAR3 expression. Am J Physiol Lung Cell Mol Physiol . 2021 Jul 1;321(1):L65-L78.
PubMed

Arun Prakash, M.D., Ph.D.
Arun Prakash completed his undergraduate training at the University of Rochester where he studied Chemistry and History and worked in the laboratory of Professor Joseph Dinnocenzo in the Chemistry Department at UofR.
He completed his MD and PhD degrees at New York University, where he completed his doctoral dissertation in the laboratory of Professor David E. Levy.
He then completed his medical internship at Santa Clara Valley Medical Center in San Jose, CA before starting his Anesthesiology training at UCSF.
He was appointed a Clinical Instructor after completing his residency training in 2009 and in 2011 was appointed Assistant Professor in Residence.
Xiaoli Tian, Ph.D.
Lab Manager
Daisuke Maruyama
T32 scholar/post-doc
Thien Doan
Staff Research Assistant
The Hellman Lab
We work very closely with members in the Hellman Lab and share space and equipment with them.
Other Labs and Groups that we work closely with at UCSF:






 c
c

Past Members
2023
Farzaneh Kianian (Visiting Researcher)
Christine Tat
2019
Qing Liu
(Graduate Student)
2018
Zinnia Finn (high school student)
Molly Carleton (undergrad)
Michelle Ullman (undergrad)
2017
Molly Carleton (undergrad)
Calvin Chan (undergrad).
2011-2012
Adam Ferris
SRA 1
Current Position: SRA 2 (UCSF Dept. of Neurology)

Shirin V. Sundar, Ph.D
Associate Specialist (2012-2014)
Currently at: Otsuka America Pharamceuticals, Inc

Brendon Owczarek
High school summer intern, 2015

Sun He, M.D.
Visiting Scholar
2015-2016


Arun Prakash
[email protected]
University of California, San Francisco
Department of Anesthesia and Perioperative Care
Zuckerberg San Francisco General Hospital
1001 Potrero Ave. Building 5, Room 3C38
San Francisco, CA 94110
FAX (415)-206-6014 Direct (415) 206-8969
BOX: 1371
If you are interested in applying for a position please send me an email with your CV. Thank you.

Our research seeks to understand the pathophysiology of lung injury in trauma patients. Our goal is to develop strategies and therapies that can help improve the outcome of severely injured trauma patients.
If you would like to help support this research program, the link to the UCSF donate page and paypal that will help fund the experiments we do is below:
(please designate that you would like your contribution to support 'the Prakash Lab' under <choose a designation> and <other>)